Scientific Expertise
Explore educational resources about the testing solutions needed to get your device to the next stage along with important administrative forms so you can be set up for success.
Explore educational resources about the testing solutions needed to get your device to the next stage along with important administrative forms so you can be set up for success.
Featured Content
Labcorp's coffee talk presentation and MD&M West 2023
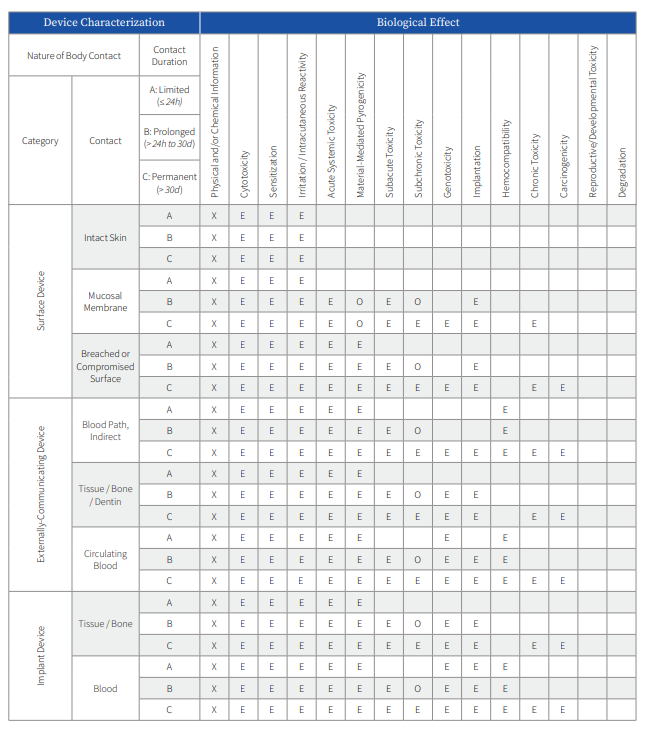
Biocompatibility endpoints required for assessment as a part of a risk based evaluation of medical device safety are provided by ISO 10993-1 and various international regulatory body standards and guidance documents. The endpoints detailed in the matrix table describe the potential risks based on the clinical contact type and duration of the medical device to the patient or clinician. Devices may fall into multiple categories based upon the various components (delivery system and implant), and it is important to consult all available applicable standards and guidance documents which may provide other specifics to the medical device biocompatibility evaluation process. Each endpoint for evaluation should be identified as a part of a biological evaluation plan (BEP).
| Acronym | Agency/Term/Organization |
|---|---|
| FDA | Food and Drug Administration |
| EPA | Environmental Protection Agency |
| ISO | International Standards Organization |
| USDA | United States Department of Agriculture |
| MSPCA | Massachusetts Society for the Prevention of Cruelty to Animals |
| AAALAC | International Association for the Accreditation of Laboratory Animal Care and Use |
| OLAW | Office for Laboratory Animal Welfare |
| DEA | Drug Enforcement Agency |
| MWRA | Massachusetts Water Resource Authority |
| EPA | Environmental Protection Agency - Hazardous Waste Division |
| MA-DEP | Department of Environmental Protection |
| MA-DPH | Massachusetts Radiation Control Program |
| NRC | Nuclear Regulatory Commission |
| CLIENTS | Medical Device, Pharmaceutical Chemical Manufacturers |